Primary alcohols can also undergo a dehydration reaction via an E2 mechanism, but with a much slower rate than secondary and tertiary alcohols. Since this reaction also removes a water molecule, chemists also call it a “dehydration reaction”. Secondary and tertiary alcohols can undergo an E1 reaction to form alkenes under acidic conditions. It’s important to note that since OH is a bad leaving group, the conversion of OH into a better leaving group is a required step in the mechanism of every alcohol substitution reaction. Tertiary alcohols only undergo S N1 reactions secondary alcohols can undergo S N1 reactions but with a slow rate and thus generally prefer to undergo S N2 reactions primary alcohols only undergo S N2 reactions. Reactions of Alcohols Substitution ReactionsĪlcohols can undergo substitution reactions to form alkyl halides. A Grignard reagent has a nucleophilic carbon that can perform attacks on various electrophiles and thus is useful in building carbon skeletons. In our case, aldehydes, ketones, and esters can all be attacked by the Grignard reagent and undergo subsequent protonation to produce alcohols. Grignard reagents are alkyl halides that are treated with magnesium. These reducing agents act as proton sources, giving their protons to the reactant. Moreover, the reactivity of LAH also allows it to reduce carboxylic acids and esters into alcohols. We can reduce aldehydes or ketones into alcohols with a reducing agent like sodium borohydride (NaBH 4) or lithium aluminum hydride (LAH). You can read more about these hydration reactions in our article on alkenes. Addition ReactionsĪlkenes can undergo hydration to produce alcohols. An S N1 reaction typically yields a tertiary alcohol, while an S N2 reaction typically yields a primary alcohol. We can produce alcohols using either an S N1 or S N2 substitution reaction. Preparation of Alcohols Substitution Reactions The high electronegativity of oxygen makes the C and H bonded to it electrophilic and thus reactive to electron-rich molecules.īut before jumping into the reactions of alcohol, let’s see how one can synthesize alcohol first. You will see many reactions that involve removing the whole OH group or just the H. The reactivity hotspot in alcohols is the OH group.
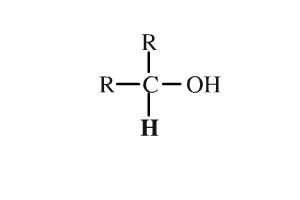
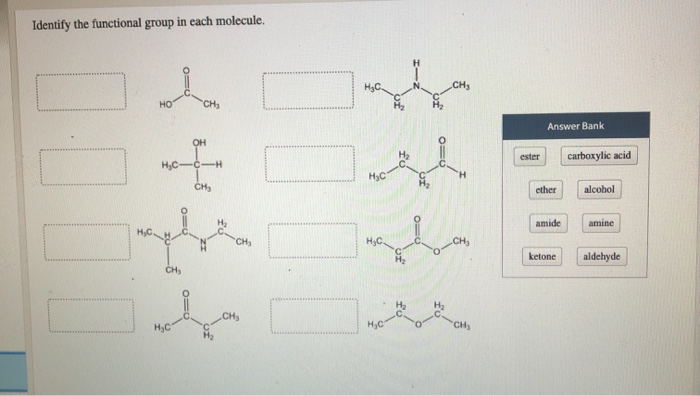
In such cases, keep the “-e” of the parent alkane.Ħ-hydroxy-3-methyloctanoic acid Alcohol Reactions General Reaction Trends If a molecule has multiple OH groups, insert prefixes like “di” or “tri” before “-ol”. Finally, change the suffix “-e” of the parent alkane into “-ol”.The OH group should get the smallest number possible. Next, assign a number to the OH group and other substituents (if there are any) to indicate their position within the molecule.First, identify the parent, which is the longest chain that contains the OH group.To name an alcohol using IUPAC nomenclature, follow these steps: IR Spectroscopy: A concentrated alcohol solution produces a broad signal around 3200 – 3600 cm -1.For example, methanol and ethanol are miscible in water, while nonanol is insoluble in water. Solubility: The solubility of an alcohol in polar solvents decreases as the size of the hydrophobic region (typically the hydrocarbon chain) of that alcohol increases.Alcohols are generally weak acids whose most acidic proton is the H of the OH. Structure: A carbon single bonded to an oxygen that’s single bonded to a hydrogen.Dopamine, a phenol Quick Facts on Alcohols
